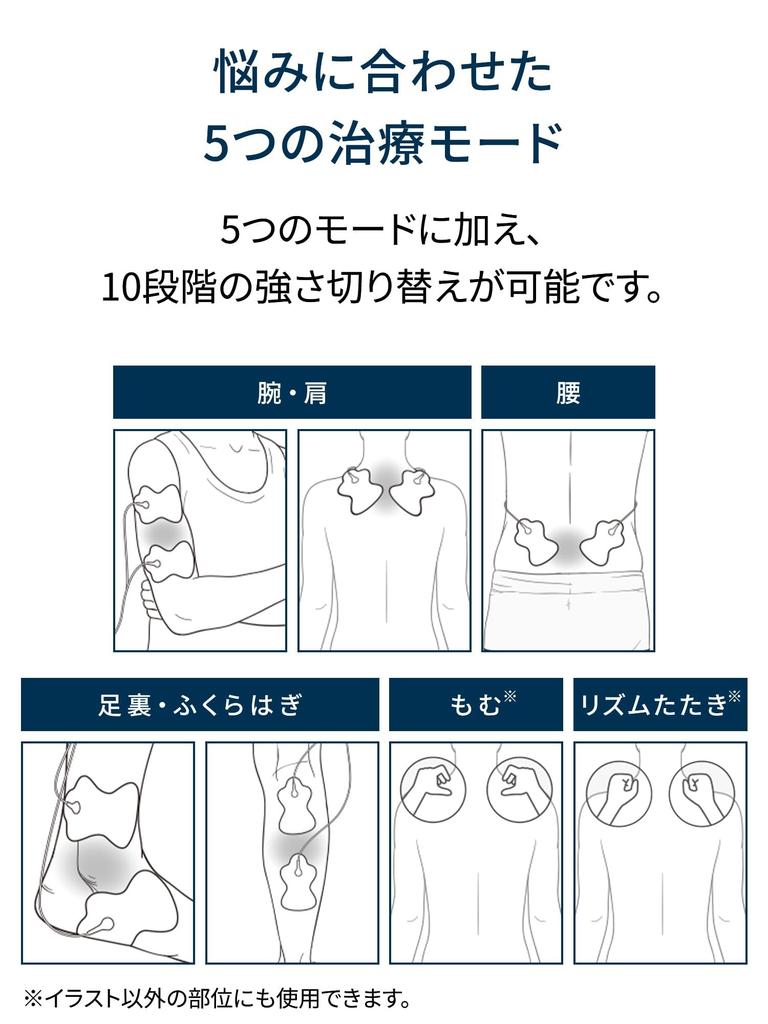
[Please refer to the instruction manual]
[Generic Name]
Home Low-Frequency Therapy Device
[Product Name]
Omron Low-Frequency Therapy Device HV-F013 Series
[Warning]
The following individuals should consult a doctor before using this product:
(1) Those currently receiving medical treatment or those experiencing any physical abnormalities
(2) Those with malignant tumors
(3) Those with cardiac or neurological abnormalities
(4) Pregnant women or those who have just given birth
(5) Those with a body temperature of 38°C or higher (fever)
(Example 1: Those experiencing severe acute symptoms (fatigue, chills, blood pressure fluctuations, etc.) 2: Weakened)
(6) People with infectious diseases
(7) People with skin sensory impairment or other skin abnormalities
(8) People with acute (painful) illnesses such as spinal fractures, sprains, or pulled muscles
(9) People requiring rest
(10) People taking medication
(11) People with abnormal blood pressure
(12) People with limb disabilities
(13) People unable to express themselves
(14) People with sensory impairment due to severe peripheral circulatory disorders such as diabetes
(15) People with low temperatures
(16) People with poor circulation
(17) People who do not see results even after using the device for a while
(18) People with skin discomfort caused by the pads
(19) People with metal embedded in their bodies
• Risk of accident or ill health.
Do not use near the heart, above the neck, on the head, inside the mouth or genitals, on skin conditions, or to treat both soles of the feet simultaneously.
Also, do not use in a way that pinches internal organs.
• Risk of accident or ill health.
Do not allow people who are unable to express themselves or who require assistance to use the device alone.
• This may result in accidents, injuries, or poor health.
Do not use with other treatment devices or more than one unit at the same time, or in combination with ointments (including spray cans).
• This may cause nausea or poor health.
Do not use for purposes other than treatment.
• This may lead to accidents, problems, or malfunctions.
Do not use if the surface of the pad is chipped or damaged.
• This may result in accidents, problems, or malfunctions.
Do not insert the electrode cord insertion plug into any location other than the electrode cord insertion port on the main unit.
• This may result in electric shock or an accident.
Do not disassemble, repair, or modify.
• This may result in fire, malfunction, or an accident.
[Contraindications/Prohibitions]
Never use this device in combination with the following types of medical electrical equipment.
(1) Implantable medical electrical devices such as pacemakers
(2) Life-sustaining medical electrical devices such as heart-lung machines
(3) Wearable medical electrical devices such as electrocardiographs
• This may cause these medical electrical devices to malfunction, resulting in serious injury or death.
[Product Specifications]
Power Supply Voltage: DC 3V (Uses two AAA alkaline batteries)
Battery Life: Approximately 3 months (based on 15 minutes of continuous use once per day)
Maximum Output Current: 6mA or less
Fundamental Frequency: Approximately 1-108Hz
Rated Output Voltage: Maximum approximately 40V
Maximum Pulse Width: 100μsec Power Consumption: Approx. 0.2W
Rated Time: 15 minutes
Usage Environment: +10 to +40°C / 30 to 85% RH (non-condensing) / 700 to 1060 hPa
Storage and Transportation Environment (after opening): 0 to +40°C / 30 to 85% RH (non-condensing)
Weight: Approx. 75g (including batteries)
Dimensions: 75mm x 70mm x 22mm
Operating Principle: Utilizing the response of nerves and muscles to low-frequency electricity, a weak pulse current is applied below the skin surface, resulting in physiological effects that can be used to treat the affected area.
Lifespan: 5 years [based on self-certification (company data)]
Composition of parts that come into direct contact with the body: Acrylic resin, glycerin, water
Sold separately: Long-Life Pad (gray), Electrode Cord Type B (white), Electrode Storage Device (white)
Accessories:
Electrode Cord Type B (white) (1 piece), Long-Life Pad HV-LLPAD-GY (gray) (1 set of 2 pieces)
・Product Name: Long-Life Pad HV-LLPAD ・Manufacturer/Distributor: Sekisui Plastics Co., Ltd. ・Manufacturer/Distributor Notification Number: 08B2X10006000007, 2 AAA alkaline batteries (trial size), Electrode Storage Device (gray) (1 piece), Instruction Manual (with Quality Warranty) (1 copy)
[Precautions for Use]
If you experience any physical or skin abnormalities such as eczema, redness, or itching, discontinue use immediately.
• Consult a doctor and follow their instructions.
If you need to change pads to a different area during treatment, be sure to turn the power off before doing so.
• Risk of severe shock.
Do not ask anyone else to change pads during treatment.
• Risk of severe shock.
Do not wear electronic devices (watches, pedometers, etc.) while receiving treatment.
• The time and numbers may become inaccurate.
If the device stops working or any abnormality occurs, turn the power off immediately to prevent accidents and contact an OMRON Customer Service Center for repairs.
• Doing so may cause accidents, problems, or malfunctions due to overheating or a short circuit.
Do not plug/unplug the cord or attach the pads with wet hands.
• Risk of electric shock or accidents.
Do not allow children to use the device or play with the device or pads. Also, do not allow them to climb on them.
• Risk of accidents, injury, poor health, or malfunctions.
Do not use in humid places such as the bathroom, or while bathing.
• Risk of severe shock.
Do not use while sleeping.
• Doing so may cause damage to the device. The pads may also stick to unexpected areas, causing discomfort.
Do not use while driving a car or other vehicle.
• Strong irritation may result in an accident or other problems.
Do not leave the pads attached to the affected area.
• Doing so may cause skin irritation.
Do not use the pads while they are in contact with metal objects such as belts or necklaces.
• Doing so may result in a strong shock.
[Name of Manufacturer/Distributor]
OMRON HEALTHCARE Co., Ltd.
53 Kunotsubo, Terato-cho, Hyuga-shi, Kyoto 617-0002
Tel: 0120-30-6606 (OMRON Customer Service Center)
[Purpose or Effects/Efficacy]
Relieves stiff shoulders, prevents atrophy of paralyzed muscles, and provides a massage effect. For general household use.
Medical Device Classification: Specially Controlled Medical Device
Medical Device Certification Number: 306AHBZX00017000
Power Supply: 3 V DC (Uses two AAA alkaline batteries)
Weight: Approximately 75 g (including batteries)